Drug Development/R&D
New medicines lead to improved patient outcomes and societal benefits, but R&D is a long and costly process. Ongoing health economics research aims to find the right balance between incentivising manufacturers to innovate and ensuring affordable access for health systems.

New Publication: Spillovers between Public and Private Sector Biomedical and Health Research
8 March 2016
OHE’s Yan Feng and Jorge Mestre-Ferrandiz have co-authored a new publication on quantifying the spillovers between public and private sector biomedical and health research and development…

New Publication: Headroom Approach to Device Development
23 February 2016
OHE’s Amanda Cole co-authors a new publication on the headroom approach, which can help assess the commercial value and viability of medical device development.
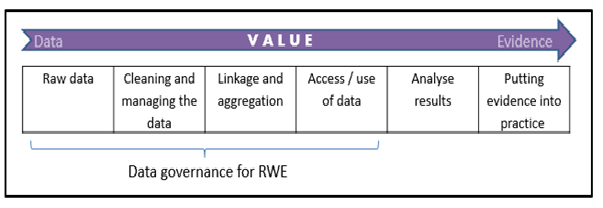
How to Manage Data Governance in an Era of Real World Evidence (RWE): Working Towards a Set of International Standards
24 November 2015
A new OHE Consulting Report assesses information governance arrangements for real-world data in eight countries, and makes recommendations towards an ideal governance framework.

Newly Published Analysis: Dementia: the R&D Landscape
16 November 2015
Just published by the World Dementia Council and OHE is an analysis which sets out the dementia R&D landscape. This analysis was undertaken by OHE for…

Multi-indication Pricing: Pros, Cons and Applicability to the UK
28 October 2015
Multi-indication pricing (MIP) involves setting a different price for each major indication approved for a medicine. We explore the feasibility of implementing MIP in the UK.
Future Cases of Dementia among People Born in 2015 & the Dementia Drug Pipeline
6 October 2015
It is estimated that one in three people born in 2015 in the UK may develop dementia, and clinical development success rates for dementia drugs are…

PriMHE Lecture: Relative Effectiveness and the Impact on Drug Development
28 May 2015
OHE’s Adrian Towse will give a lecture at the Medical University of Vienna on relative effectiveness and the impact on drug development.

OHE at ISPOR Philadelphia Part 2: Risk Sharing, Medical Breakthroughs, Early Access and Drug Development Costs
12 May 2015
This post is the second of a two part summary of OHE’s scheduled activity at ISPOR Philadelphia 2015.

OHE Publishes in the Journal of Comparative Effectiveness Research: Future Expectations of Evidence of Relative Effectiveness in Europe and Comparative Effectiveness in the USA in 2020
15 April 2015
Articles co-authored by Adrian Towse, Martina Garau, Donna Messner, and Penny Mohr exploring future expectations of drug relative effectiveness in Europe and comparative effectiveness in the…