Japan’s health technology assessment (HTA) system uses algorithmic drug pricing and cost-effectiveness analysis to manage rising healthcare costs while maintaining universal access and innovation.
The Health Care System in Japan
Like the precise turning of a shōgi piece, Japan’s health system moves strategically, delivering exceptional care as it increasingly focuses on managing costs.
Established in 1961, Japan’s mandatory, multi-payer social insurance system provides universal health coverage for its citizens.
All residents must be covered, so most salaried workers and their dependents are part of Employees’ Health Insurance schemes, while the self-employed, retirees, and others join National Health Insurance through their municipality.
Financing comes from income-linked premiums, local and national government contributions, and patient cost-sharing. Out-of-pocket payments are typically 30% for working-age adults, with reduced rates for children and older adults and monthly caps that protect households from catastrophic costs.
Prices and payment are also tightly regulated. A single national fee schedule sets fees for virtually all services. Patients have free choice of providers, mainly privately operated, non-profit hospitals and clinics alongside public institutions. Long-term care is covered under a separate social insurance program.
An excellent system under pressure
This system achieves excellent health outcomes, such as high life expectancy, while costs continue to rise due to:
- Demographic challenges – An ageing population is putting pressure on the system, as nearly a third of citizens are over 65. Coupled with population decline due to a low birth rate, this demographic shift is increasing demand for health services while the tax base is shrinking.
- Health care inefficiency – The health care system is fragmented and hospital-centric. Financial incentives encourage long hospital stays and extensive treatment, making Japan’s average inpatient duration the longest in the world. Limited primary care and reliance on prescriptions also drive polypharmacy and higher costs.
Intensifying this pressure on healthcare costs is the general financial crisis in Japan. Japan has one of the highest public debt burdens among developed economies – total debt is projected to reach 235% of GDP this year. There is also enormous political pressure related to increasing expenditure, for example, on defence, and to stimulate economic growth, while also cutting taxes to combat the high cost of living.
Sustainability of the healthcare system has become a major concern, and according to the Ministry of Finance, overall medical and nursing costs have doubled over the past twenty years, far outstripping economic growth.
Attempts at reform to healthcare financing have received political pushback, and the government has instead turned to cost containment, with drug prices absorbing many of the reductions – while less than 10% of the country’s social security budget is spent on medicines, 70% of budget reductions are due to drug price cuts.
The Health Technology Assessment (HTA) system
Japan has a centralised pricing system which sets a single national price for all reimbursed drugs; all drugs that receive a marketing authorisation by the regulatory authority are automatically reimbursed within 60-90 days. It’s a ‘price setter’ market, where initial pricing is determined by the Ministry of Health, Labour and Welfare (MHLW) using an algorithmic approach. The different pricing approaches are summarised in Figure 1. Price premiums can be awarded to drugs that meet certain criteria, including being ‘innovative’, for example, by being judged to have greater efficacy than the comparator. Once a price has been determined, the MHLW propose that price to the Central Social Insurance Medical Council (Chuikyo) for decision-making.
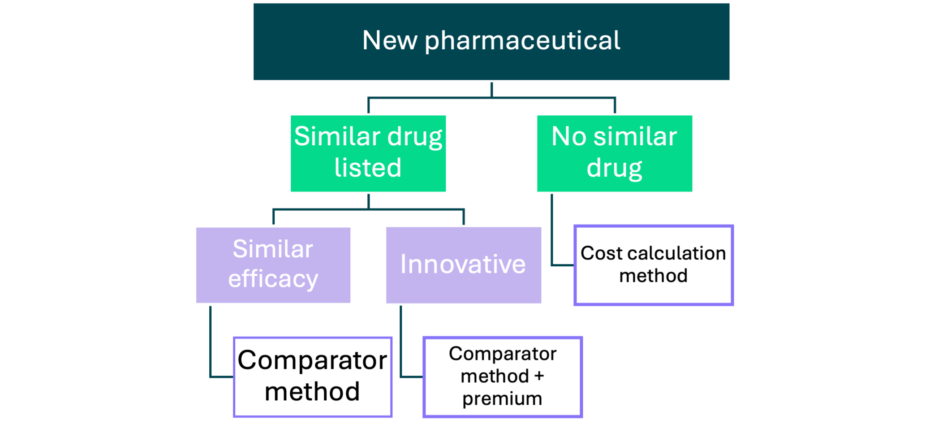
Pricing of new pharmaceuticals
Once drugs are listed and available to patients, Japan employs a unique ‘re-pricing’ process to reduce drug prices under various circumstances, including via cost-effectiveness assessment (CEA).
Establishing the CEA system
Unlike other HTA systems, the Japanese process does not determine coverage decisions. Instead, CEA repricing was implemented to evaluate and adjust the premiums granted to drugs that were considered to be innovative at the time of initial price setting, as well as the operating profit under the cost calculation method. Following several HTA pilots that started in 2016, the CEA price adjustment system was established in 2019.
The CEA system is overseen by the Chuikyo’s subcommittee on cost-effectiveness, while the technical assessment is conducted by the Center for Outcomes Research and Economic Evaluation (C2H), often referred to as the “Independent Expert Organisation”.
There are five categories to determine if a drug or device is subject to CEA:
| HI | If the peak annual sales of a product listed after full implementation of CEA is estimated to be over ¥10billion (US$68million); |
| H2 | If peak annual sales of a product listed after full implementation of CEA are estimated to be between ¥5-10 billion (US$34 million – US$68 million) they may be considered depending on capacity |
| H3 | If a product listed after full implementation of CEA is perceived by the Chuikyo to have a significantly high price |
| H4 | If annual sales of a product listed before full implementation of CEA exceed >¥100billion (US$679 million) or have a significantly high price |
| H5 | If the price of a product has been determined using a comparator that was classified under H1-H4 |
Source: Full scale introduction of cost-effectiveness evaluations in Japan (2019) Medical Economics Division, Health Insurance Bureau, Ministry of Health, Labour and Welfare (MHLW), Japan https://c2h.niph.go.jp/tools/system/overview_en.pdf
For newly listed drugs subject to CEA, the company must submit a dossier within nine months of launch. The process involves independent expert assessments and discussion appraisals by the Chuikyo CEA subcommittee.
How CEA works
After calculating the incremental cost-effectiveness ratio (ICER) in line with C2H method guide, an algorithmic approach is used in which the ICER determines the size of the price adjustment.
Specifically, a tiered ICER threshold model determines the level of price adjustment for the drug.This threshold can be modified for drugs with ‘special considerations’, namely rare, paediatric or cancer indications. The price adjustment is applied to the premium portion of the price, or the operating profit portion for drugs priced under the cost calculation method, according to these thresholds:
| Threshold(FX: 151.8¥ = $1USD) | Repricing rate (where, for example,1.0 = no repricing, 0.5 = price halved) | ||
| Standard | Special consideration | Operating profit | Premium |
| < ¥5 million (US$$33,000) | < ¥7.5 million (US$49,400) | 1.0 | 1.0 |
| ¥5 – ¥7.5 million (US$33,000 – US$49,400) | ¥7.5 – ¥11.25 million (US$49,400 – US$74,200) | 0.87 | 0.7 |
| ¥7.5 – ¥10 million (US$49,400 – US$65,900) | ¥11.25 – ¥15 million (US$74,200 – US$98,800) | 0.63 | 0.4 |
| > ¥10 million (US65,900) | > ¥15 million (US$98,800) | 0.5 | 0.1 |
There is a maximum price reduction capped between 10-15% of the drug’s total price, depending on the size of the initial premium. The method also makes sure that the adjusted price does not fall below the ‘cost-effective price’ calculated at the baseline threshold of ¥5 million (US$33,000) per QALY. It is also possible for the CEA to allow modest price increases for products deemed dominant to the relevant comparator, although this is seldom used.
Current and emerging challenges
There are a number of challenges related to Japan’s HTA system. Firstly, unlike many other HTA systems, there is a lack of a formal deliberative appraisal process to place the technical cost-effectiveness analysis in a broader social, ethical, and policy context. While the Chuikyo reviews price adjustment recommendations, deliberations appear more limited than appraisal committees in HTA systems with a broader remit and are primarily focused on price adjustments. This challenge is compounded by lack of input from patients, healthcare providers, and industry stakeholders, which are important for maintaining transparency and trust in the system.
There is an ongoing discussion about expanding the CEA system in Japan, however, there appear to be capacity issues in the current system. To date, a relatively low number of drugs have been selected for CEA since 2019, reflecting a shortage of domestic experts trained in HTA methodologies.
With respect to expanding CEA, recent proposals (e.g., by the Ministry of Finance) have suggested extending CEA price adjustments to the total price of products and additionally to products without premiums. In August 2024, an “ad hoc special rule” was applied to the Alzheimer’s Disease treatment Leqembi using a new process that compared the difference between Leqembi prices with the calculated cost-effective price at the baseline threshold. That difference was used to determine a reduction to total price, although capped at a maximum of 15%. There’s a real possibility that this could be considered a pilot to pave the way for the ad-hoc rule to become more standard.
However, care will need to be taken to address potential policy mismatches between the CEA’s original intent and its proposed expansion. Repricing approaches could theoretically result in a price below the comparator, leading to unintended consequences such as reduced incentives for companies to generate evidence of added benefit or further disinvestment in Japan-focused R&D. While Japan’s pricing system has successfully controlled government pharmaceutical expenditure, expansion must be carefully calibrated to avoid policy incoherence.
Next steps
Japan’s CEA system may have begun modestly, but the debate over its future is intensifying.
A six-month technical discussion and ‘third-party verification’ of existing CEA results is commencing in early 2026 with a report scheduled to be submitted to the Chuikyo CEA subcommittee in September 2026. At that time, the Chuikyo will consider the outcome of the report and its implications for their intended expansion of CEA – both in terms of eligible products and the potential shift from premium-based to total price adjustments.
Beyond expanding the scope of CEA, there have also been proposals to use CEA for reimbursement decision-making, referencing the utilisation of HTA in the UK and Germany for this purpose. While this more dramatic change in approach is currently off the table in terms of implementation, discussion is ongoing.
An additional consideration is that, at the time of writing this insight, Japan has been identified as one of the countries to be included in the basket for the US Most Favoured Nation (MFN) policy. Although significant uncertainty remains, MFN could bring significant implications for the Japanese pharmaceutical market, potentially affecting pricing and access to innovative medicines, and the overall exposure of Japan’s system to global pricing pressures.
Japan’s efforts to balance rising healthcare costs with innovation highlight a delicate challenge. The country’s demographic pressures and need to contain costs makes careful, evidence-based policy more important than ever. However, it’s essential such measures to do not stifle innovation or restrict patient access.






