Data exclusivity is critical to ensuring that biosimilars achieve an appropriate balance between increasing market competition and encouraging innovation. The history of ‘traditional generics’ is a…
Data exclusivity is critical to ensuring that biosimilars achieve an appropriate balance between increasing market competition and encouraging innovation. The history of ‘traditional generics’ is a poor guide; biosimilars possess critically different characteristics. In the seminar, Prof Grabowski explored these differences and presented his analyses of data exclusivity requirements for biologics/biosimilars.
This is the third in our series of posts based on the OHE seminar and summarises the remarks of Prof Henry Grabowski of Duke University.
Data exclusivity is the period of time after a drug is approved before a biosimilar product can enter the market based on an abbreviated regulatory filing that relies at least in part on the innovator’s safety and efficacy data. For new chemical entities, data exclusivity is 5 years in the US and 10 years in Europe. The question is whether biologics should have a longer data exclusivity period because of the narrowness of biologic patents, which are primarily process or formulation patents. Biosimilars may be different enough to not infringe on the innovator’s patent, yet similar enough to qualify for an abbreviated regulatory approval process. Data exclusivity, then, provides important protection and incentives.
In determining the appropriate length of a data exclusivity period for biologics, small molecule drugs are not a good guide. Biologics are characterised by higher discovery costs, longer development times and higher capital investment in manufacturing plants. Development times for biologics, moreover, have more than doubled in the past 25 years and development progression is fraught with uncertainty because it often relies on venture capital.
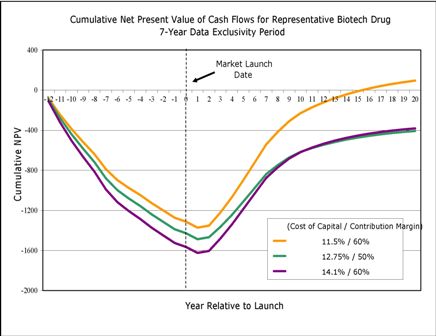
Prof Henry Grabowski’s presentation focused on approaches to analysing a ‘reasonable’ data exclusivity period for biologics He presented a recent analysis for a portfolio of biologic products, using a series of assumptions about sales, production costs, obsolescence and cost to capital that he considers conservative. Preliminary results showed that breakeven times range from 12.9 to 16.2 years for biologics, without competition from biosimilars.
In estimating the potential effect of biosimilars on breakeven times and incentives for innovation, Prof Grabowski noted that price competition will be substantially less for biosimilars than for generics because of two factors. On the supply side, development costs for biosimilars are much higher than for generics; on the demand side, uptake of biosimilars will be affected by payer and provider uncertainty about interchangeability. Overall, market penetration and price erosion will be slower for biosimilars than for generics.
Prof Grabowski pointed out that several attempts have been made to estimate the extent of the impact of biosimilars in the US market. This includes his own analyses based on ‘complex small molecules’ that experienced generic competition in the US in the late 1990s to mid 2000s. These molecules, according to the analysis, lost about 40% of market share over two years, compared with 80% for drugs without complex characteristics. Based on a simulation exercise that focuses on the cost of capital, contribution margins, biosimilar share and price discounts as well as the data exclusivity period, Prof Grabowski suggested that a data exclusivity period of at least 10 to 14 years is needed to encourage continued innovation in biologics. (These analyses appear in great detail in the publication.)
Editor’s note: The US Biologics Price Competition and Innovation Act of 2009, enacted since this conference, provides for an abbreviated approval process for biosimilars, a 12-year data exclusivity period and FDA determination of ‘interchangeablility’.
Henry G. Grabowski is Professor Emeritus and Director of the Program in Pharmaceutical Health Economics, Duke University.
Publication now available for download: Mattison, N., Mestre-Ferrandiz, J. and Towse, A. eds (2010) Biosimilars: How much entry and price competition will result? London: Office of Health Economics.