Health Technology Assessment (HTA)
Health Technology Assessment (HTA) is a multidisciplinary process that uses explicit methods to determine a health technology’s value for healthcare decision-making. Attitudes towards HTA vary globally, with some countries using a variety of methods and analyses and others hardly any.

New OHE Publications: An EQ-5D-5L Value Set for England
22 March 2016
Two newly published OHE Research Papers report the methods and findings of the ‘EQ-5D-5L value set for England’ study.

Recently published: Are Wider Societal Effects Considered in Healthcare Decision-making?
29 February 2016
Health interventions can produce economic gains for patients and national economies. In practice, however, the health-wealth link is rarely considered.

Just Published: OHE Report on Trends in Scottish Medicines Committee Advice Decisions
25 February 2016
An analysis in the trends in Scottish Medicines Committee (SMC) advice decisions for medicines for the period October 2009 to September 2015 has just been published…

New Publication: Headroom Approach to Device Development
23 February 2016
OHE’s Amanda Cole co-authors a new publication on the headroom approach, which can help assess the commercial value and viability of medical device development.

New ISPOR Task Force Report on Multiple Criteria Decision Analysis
9 February 2016
Professor Nancy Devlin co-chaired the ISPOR task force on uses of MCDA in health care decision making. The first report from the task force has just…
Extrapolation from Progression-Free Survival to Overall Survival in Oncology
1 December 2016
This Research Paper reports on a literature review of trials using PFS as a surrogate for OS in oncology between 2012 and 2016; a workshop and…

Applying a Multi-criteria Decision Analysis (MCDA) Approach to Elicit Stakeholders’ Preferences in Italy. The Case of Obinutuzumab for Rituximab-Refractory Indolent Non-Hodgkin Lymphoma (iNHL)
1 December 2016
Published paper using MCDA to obtain preferences on decision criteria across three stakeholder groups (patients, clinicians and payers) in Italy and to assess the value of obinutuzumab for rituximab-refractory iNHL.
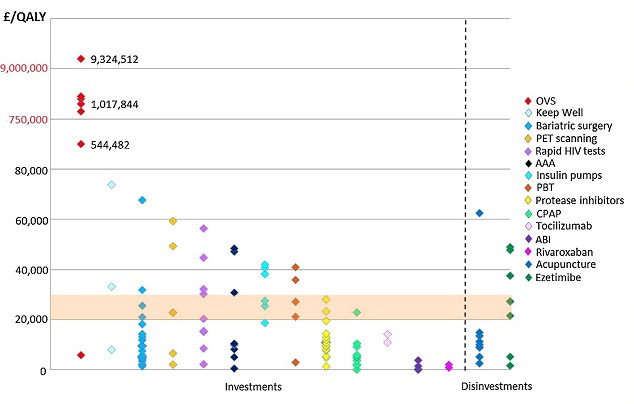
Seminar at Glasgow Caledonian University: Local Health Care Expenditure Plans and Their Opportunity Costs
11 January 2016
OHE’s Sarah Karlsberg Schaffer presented at the Glasgow Health Economics Seminar Series the results of a paper on the value of the cost-effectiveness threshold in Scotland.
Uncertainty and Risk in HTA Decision Making
1 November 2016
The quality of decision-making in key public sector bodies dealing with resource allocation is a major determinant of their efficiency. One of the most difficult and contentious areas of decision-making is the way that uncertainty is dealt with.