Risk-Sharing Arrangements

OHE at AES 2016: Early Access Schemes, Equity Adjusted QALYs, Multi-indication Pricing and Formulary Development
2 August 2016
The Spanish Health Economics Association (AES) Conference was held at the University of Murcia, 15-18th June 2016. The topic of the meeting was “Reforms under the…

New OHE Consulting Report on Shaping Research Agenda to Estimate Cost-effectiveness Thresholds for Decision Making
18 April 2016
OHE publishes new Consulting Report on shaping the research agenda to estimate relevant cost-effectiveness thresholds for Health Technology Assessment decision making.

Spotlight on OHE: Beyond QALYs, Managed Entry Agreements, Business Models for Antibiotics, and the UK Market for Medicines.
12 April 2016
The blog provides links to slide sets for presentations relating to going ‘beyond QALYs’ when valuing health care interventions, managed entry agreements within the UK pharmaceutical…
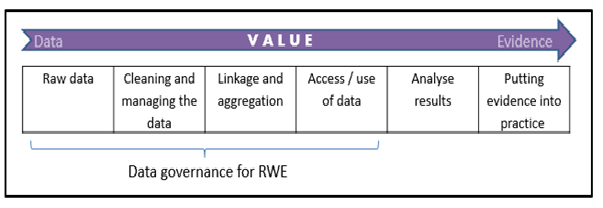
How to Manage Data Governance in an Era of Real World Evidence (RWE): Working Towards a Set of International Standards
24 November 2015
A new OHE Consulting Report assesses information governance arrangements for real-world data in eight countries, and makes recommendations towards an ideal governance framework.

New Publication: Risk-Sharing Agreements Between Pharmaceutical Manufacturers and Payers
25 September 2015
Recently published is a study by a research team including OHE’s Adrian Towse which examines prospects for risk-sharing agreements between pharmaceutical manufacturers and payers in the…

OHE at ISPOR Philadelphia: Presentations and Posters
4 June 2015
This blog contains a selection of the posters and presentations given by OHE at ISPOR Philadelphia in May 2015.

OHE at ISPOR Philadelphia Part 2: Risk Sharing, Medical Breakthroughs, Early Access and Drug Development Costs
12 May 2015
This post is the second of a two part summary of OHE’s scheduled activity at ISPOR Philadelphia 2015.

Spotlight on OHE: Adaptive Pathways, Economic Evaluation, Stated Preferences and Life-cycle Price Modelling
5 November 2014
Medicines Adaptive Pathways: A Practical Strategy to Improve Patient Access to Medicines? In September 2014 the Centre for Innovation in Regulatory Science held a workshop…

OHE at ISPOR Europe 2: Conceptual and Practical Challenges in Judging Value
29 October 2014
ISPOR’s 17th Annual European Congress is scheduled for 8-12 November 2014 in Amsterdam. This is the second of two blog posts that outlines OHE’s involvement. (The…