Making Outcome-based payments a reality in the NHS authored by The Office of Health Economics, RAND Europe, King’s College London, and Cancer Research UK, and commissioned…
Making Outcome-based payments a reality in the NHS authored by The Office of Health Economics, RAND Europe, King’s College London, and Cancer Research UK, and commissioned by Cancer Research UK in partnership with Greater Manchester Health & Social Care Partnership, explores alternative pricing models and the implications of outcome-based payment schemes for improving access to cost-effective cancer drugs.
One way to speed up access to new medicines, as suggested by the 2016 Accelerated Access Review, is for there to be more flexible pricing mechanisms. A payment model that is gaining traction is outcome-based payment (OBP), whereby the price paid for the drug is linked to the real-world outcomes it achieves for patients
The Office of Health Economics, RAND Europe and King’s College London were awarded a research grant from Cancer Research UK in partnership with Greater Manchester Health & Social Care Partnership (GMHSCP) with the following objectives:
- Produce a body of evidence that can underpin criteria to evaluate the real-world clinical benefit of a new cancer drug.
- Define the criteria, in a manner which can provide a benchmark for future outcome-based payment schemes for cancer drugs with cancer-specific measures.
- Be pragmatic and produce evidence that is transferable to a clinical setting in the NHS, and can be used to improve patient outcomes and drive value for the health service – taking into consideration the existing capabilities as well as the potential for future indicators to be developed.
The research report has just been published and can be found here. The research team explored the current funding landscape in England, with a focus on the role of patient outcomes in the drug approval process, as well as current patient access and pricing arrangements. A review of the relevant literature provides a synthesis of the available evidence on existing OBP schemes internationally and identifies the key steps required for the implementation of OBP in England.
The research went on to explore which outcomes cancer patients value most, as reported in the literature on cancer treatment outcome measures (including clinical outcomes, patient reported outcomes and patient experience measures), and using two focus groups with patients and an online survey with patients and carers. The figure below illustrates the full spectrum of outcome types. Among these, the treatment outcomes that people affected by cancer consider most important are: survival; disease progression, relapse or recurrence; long-term side effects; and return to normal activities. It is these treatment outcomes that should inform an OBP approach.
Interviews with clinicians, commissioners of cancer services, representatives of the pharmaceutical industry and academic experts provided additional evidence on OBP schemes as well as some initial insights on the data capabilities within the NHS. Based on these findings and analyses the report makes several recommendations for taking forward OBP for cancer medicines both within Greater Manchester (with its devolved responsibility for NHS and social care) and at a national level.
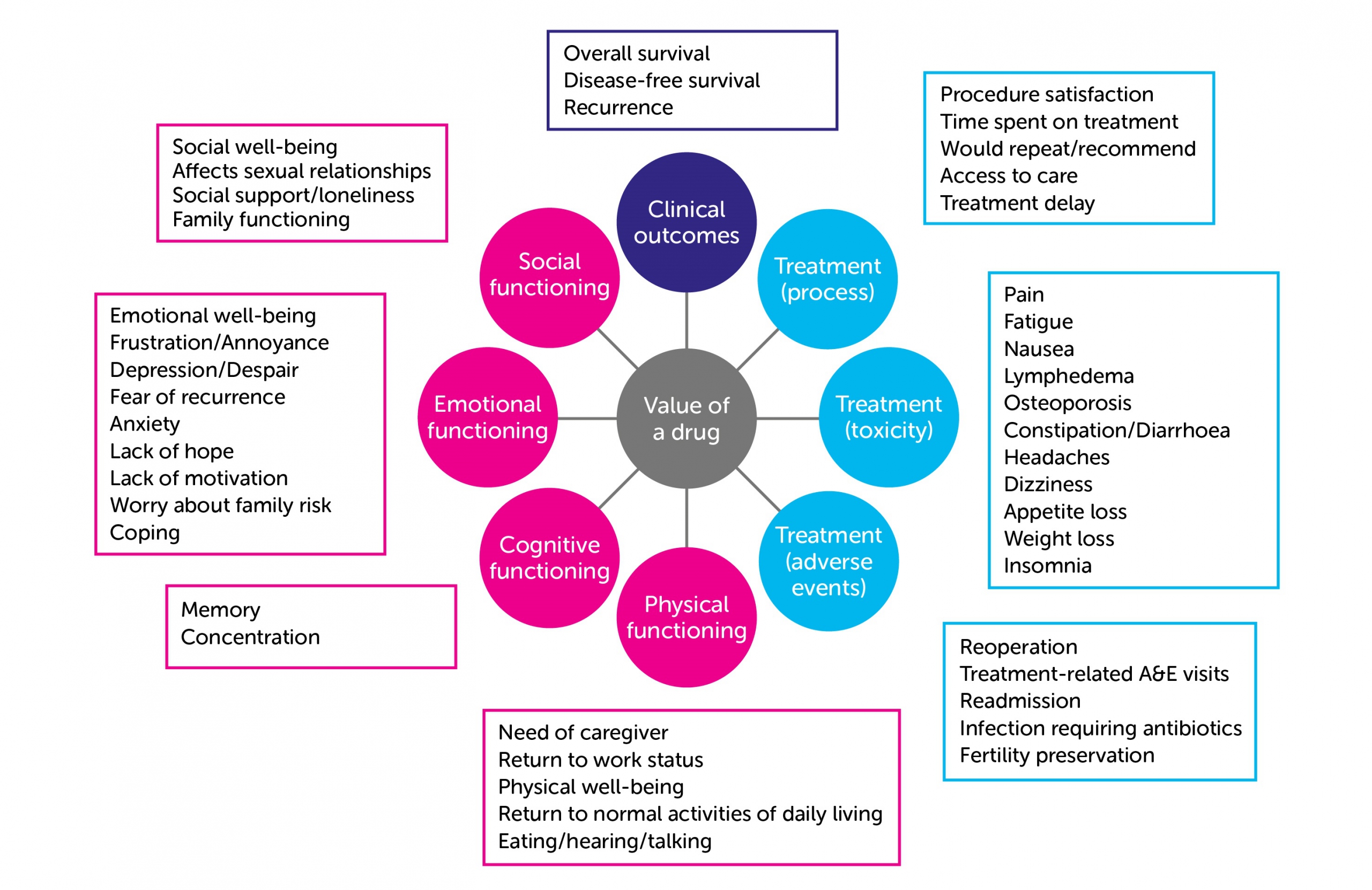
Source: Making Outcome-based payments a reality in the NHS report
Emma Greenwood (Director of Policy and Public Affairs, Cancer Research UK), Richard Preece (Executive Lead for Quality, GMHSCP), and Mike Thorpe (Patient Representative with Greater Manchester Cancer & Cancer Research UK) said of the report:
“OBP schemes have existed in the NHS previously, but they have not been used systematically, in part due to a lack of consensus between all the relevant parties. We’re pleased to have brought together a range of stakeholders – including government, NHS England, arm’s-length bodies, the pharmaceutical industry, and crucially people affected by cancer – to develop a shared vision on this topic for the first time.”
Cole, A., Cubi-Molla, P., Pollard, J., Sim, D., Sullivan, R., Sussex, J. and Lorgelly, P., 2019. Making Outcome-Based Payment a Reality in the NHS. OHE, RAND Europe, KCL and Cancer Research UK Research Paper. Available at: https://www.ohe.org/publications/making-outcome-based-payment-reality-nhs
Cole, A., Towse, A., Lorgelly, P., and Sullivan, R., 2018. Economics of Innovative Payment Models Compared with Single Pricing of Pharmaceuticals. OHE Research Paper 18/04. RePEc.
Lorgelly, P.K. and Neri, M., 2018. Survivorship burden for individuals, households and society: Estimates and methodology. Journal of Cancer Policy, 15, pp.113–117. DOI.
Cubi-Molla, P., Mott, D., Shah, K., Herdman, M., Summers, Y. and Devlin, N. (2018). Quality of Life in Long-term Cancer Survivors: Implications for Future Health Technology Assessments in Oncology. OHE Consulting Report. RePEc.
Towse, A. and Garrison, L., 2017. Value assessment in precision cancer medicine. Journal of Cancer Policy, 11, pp.48–53. DOI.