The business model for antibiotics is broken. We looked at other industries facing conceptually similar challenges – particularly when the incentive for R&D is delinked from…
The business model for antibiotics is broken. We looked at other industries facing conceptually similar challenges – particularly when the incentive for R&D is delinked from the volume of sales.
- Research: There is no globally and regionally managed consolidated focus and funding in R&D of antibiotics.
- Development: There has been a dramatic reduction of investment in R&D of antibiotics.
- Commercial: The traditional price/volume commercial model exacerbates resistance building.
Building on the experiences and insights from these other industries, along with learnings from all the work so far on antibiotics, our proposed solutions to address these three main topic areas for antibiotics are as follows:
- Ensure a global treaty or framework agreement (as seen in the Energy Industry) across all key antibiotic developers and providers to support improvements in appropriate access and use globally.
- Creation of a Global Antibiotic Public-Private Partnership (GAPPP), involving private companies, academic institutions and public bodies. There is a need for a an entity that pools resources, science and compounds; one that is sustainable and independent with focus on the research and early development of antibiotics in line with predefined public health-need profiles. This entity should find a way to open up the science to all working in this area.
2. Addressing ways to reverse reduction of return on investment of R&D:
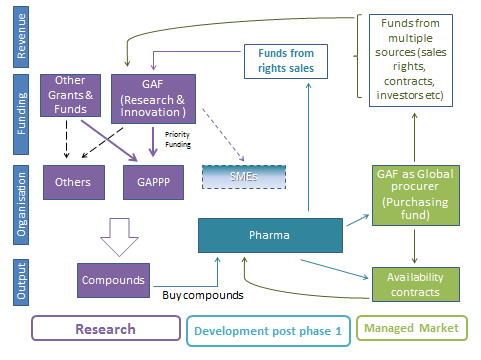
- Create a Global Antibotic Fund (GAF), which is independent and self-sustaining. The research indicates that to bring about a global ecosystem change then as a priority the GAF should provide monetary support to the new entity of the GAPPP. This Fund would be accessible to multiple companies, academic institutions and public bodies, in order to fund the appropriate research, early development and good stewardship of antibiotics.
- It is important that this GAF would work alongside the major national specific funds, and would be set up as an overarching ‘no strings attached’ fund to bring together all the small existing funds in this area i.e. failure of a particular piece of work would not require payback of the funds/grants.
- Additionally learnings indicate that there is a need to ensure self-sustainability and longevity of the GAF, therefore, as well as being a pooling of all the global small antibiotic research funds, the GAF would need to put mechanisms in place to build the additional finances needed.
- Implementing ‘Service Availability/Option of use’ contracts (as seen in other industries such as defence) with key customers such as governments. In these contracts Innovators of new antibiotics would not be rewarded with the traditional ‘price x volume’ model; instead the antibiotic would sit ready for when the surveillance indicates the new antibiotic is needed and rapid deployment and delivery is then put in place. Companies/Organisations would focus more on delivering this overall package – the product, the resources and the associated service. Governments would pay an annual insurance type ‘service-availability’ fee/premium to ensure provision of this overall package.
- Explore setting up an independent global third party brokering and procurement entity, between the provider of the product and the key customers. This Third Party would broker the ‘Availability/Option to Use’ annual contracts on behalf of all of the antibiotic developers, as well as procuring and distributing the antibiotics when the surveillance signals indicate they are needed.
All the finances from these various models combine together and feed finances a) back in to the Global Antibiotic Fund, b) back to the Pharma companies directly, c) back to the Academic institutions and SMEs that have developed the antibiotics as well as d) back to the external investors.
Figure below shows a possibility of how several mechanisms could be combined together.
This work can be seen as complementing OHE’s past analyses looking at the different types of incentives that might increase returns to antibiotics’ R&D. Access this work here.