A new OHE Consulting Report assesses information governance arrangements for real-world data in eight countries, and makes recommendations towards an ideal governance framework. Real world data…
A new OHE Consulting Report assesses information governance arrangements for real-world data in eight countries, and makes recommendations towards an ideal governance framework.
Real world data (RWD) is a term that is becoming increasingly widespread in health economics and health services research – with good reason. RWD is information that can be used for health care decision-making which is collected outside of an experimental clinical trial setting. RWD therefore has the benefit of reflecting outcomes in the “real-world”, i.e. routine clinical practice.
The environment for collecting data alongside clinical practice and using it to support decision-making by regulators and payers is changing. Regulatory flexibilities and increasing public pressure to facilitate earlier access to medicines (e.g. through adaptive pathways or early access schemes) means that regulators are increasingly monitoring benefits and risks throughout a medicine’s lifecycle; RWD is essential to support this.
Payers are similarly being challenged to conduct earlier value assessments under greater uncertainty, and re-visiting assessments as further RWD is collected. Moreover, managed entry agreements and arrangements for performance-based payments require data to be collected alongside clinical practice. These themes are central to the Accelerated Access Review currently being conducted by the UK’s Department of Health.
Supporting all of these developments is the improving infrastructure across the globe for data collection, for example through electronic health records. However, in order to utilise and derive value from RWD, appropriate and facilitative information governance is essential. RWD becomes real world evidence (RWE) – what is really of value to stakeholders – after a series of activities which facilitate the transformation of raw data into analysis and results. Robust and proportionate data governance, applied at each step, is essential in realising the value of RWD.
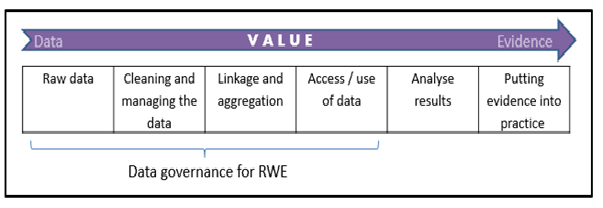
OHE Consulting has just published a Consulting Report, commissioned by Lilly, which assesses the core governance arrangements for how RWD (both routinely-collected and de-novo) is accessed or generated, and used credibly to provide evidence in eight countries: the UK, France, Italy, Sweden, Germany, the Netherlands, Australia and the U.S. In our recommendations we focus on the first four steps of the value chain set out in Figure 1. By analysing the current state-of-play in each country, we propose an aspirational governance framework that could guide the management of data access and use, and facilitate constructive interactions among the relevant stakeholders.
Governance arrangements for health data have at their core the need to balance public and privacy interests, of advancing our understanding of medical treatments through evaluation and research, on the one hand, and protecting individuals’ privacy, on the other. Different countries have different ways of addressing this aim. In most cases the legal framework in place to protect patient data is not completely prescriptive, making a clear governance framework all the more important.
We find that appropriate and facilitative information governance, along with public trust, is key to realising the benefits of scientific research. Along with sophisticated and transparent approaches to data linkage, patient consent has a key role to play, alongside mechanisms for anonymisation and authorisation. For example, where data collection is on a routine basis across a large patient cohort, an opt-out system of patient consent may be appropriate. Data can be de-identified by removing any personally identifiable information and replacing the unique patient identifier with a pseudonym.
By outlining the individual aspects and actions of an ideal framework for data governance, the authors assess the extent to which each of the countries analysed currently meets these criteria. The resulting set of recommendations work towards setting out international standards for a more facilitative environment for the transformation of RWD into RWE.
Access the full report here.
For more information contact Amanda Cole at OHE.